This Hits Home
Robert Baric, DC, BCN | When I watched my daughter grow through her stages of development, she was an outlier. She learned to ride her bike without training wheels at 2 and was able to be independent long before her peers. As she continued to grow, she gravitated to athletics, specifically swimming, becoming a nationally ranked swimmer sought after by D1 schools. She was on her way to the moon. But during this time, she was having incidental “head bumps”. Were they official concussions, no, but she definitely had symptoms for several days. Over time she started to experience many of the systemic symptoms of a TBI including anxiety, attention and sleep issues, GI issues etc.
Were these connected or random happenings?
When investigated by her doctors the frequent answer, “we do not know why the symptoms started but here is a medication if you choose to take it”. As a result of these secondary symptoms her life was now being directed by these life altering symptoms, preventing her from reaching her full potential.
Sadly, I have seen this story play out time after time. “When did your symptoms start”, I ask? And the individual responds with how long they have been struggling with them. I then ask, “tell me about your head bump two years prior to your symptoms starting”. Often, I get a very quizzical look, and an answer similar to,
“I was in an auto accident, but I was not hurt.”
Is it sports, a fall in the kitchen or a motor vehicle accident?
The brain cannot distinguish between the different injuries, and it responds in the same way to each head hit. Energy metabolism becomes disrupted, membrane potentials can no longer be maintained, and the brain’s detoxification system begins to fail. As a result, the body’s systems start to break down. The glymphatic system slows or stops functioning, as if the trash service has been on strike for weeks. All of the brain’s metabolic waste builds up, leading to significant problems.
This scenario unfolds multiple times a week in my offices, and now providers are left trying to manage a complicated situation often with disconnected system failures.
The best analogy I can offer is this: imagine you’re on vacation for two weeks, and during that time, the power goes out in your house. The food in your refrigerator spoils, mold grows, and the freezer items melt, leaking onto the floor and causing it to warp. With no AC, mold spreads throughout the house—what a disaster. Now, picture having a backup generator that kicks in automatically, keeping these vital appliances running until the power comes back. The problem is that you cannot move out; you’re stuck living in this damaged home—your body—for the rest of your life.
Introducing Concussion911, your brain’s backup generator. In the short term, you can recover quicker with an earlier return to your life! In the long term it protects your brain from the decay, damage, and mold that can disrupt your life, often preventing you from reaching your full potential.
Introduction
Every year, up to 60 million people suffer from traumatic brain injuries, including concussions, leading to staggering numbers: 27.16 million new cases, 49 million living with ongoing trauma, and a devastating 7 million years lost to disability globally (119).
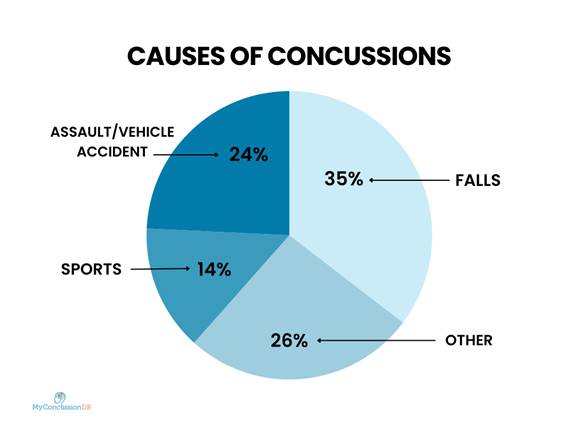
Shockingly, only 1% of all suspected concussions are reported (1). The vast majority of reported concussions occur outside of sports, with falls, assaults, and motor vehicle incidents combined responsible for the majority, accounting for more than 60% of cases (1). In contrast, sports-related concussions make up only a small fraction, around 14% (2). The consequences of these unreported and unaddressed concussions are dire on both an individual and global scale. In a study that followed 285 concussion patients using international sport concussion criteria, fewer than one-third of individuals with concussions fully recovered without intervention (3). Additionally, those who experienced symptoms for more than three years showed no signs of further recovery. While sports concussions are highly visible, it is essential to recognize that concussions can happen to anyone, anywhere.
It is clear in current research that dysfunction post-concussion is not limited to the brain; rather, concussions have a profound impact on multiple body systems, often leading to long-term health issues (4). These chronic sequelae cause severe long-term impacts, including cognitive impairments affecting academic/life performance, cardiac dysregulation, gastrointestinal dysfunction, hormonal dysregulation, decreased performance capacity in sports and other activities, mood changes including anxiety and depression often leading to interpersonal conflict, and neurodegenerative disease including dementia, Alzheimer’s and encephalopathy (5,6). Each concussion adds to the damage from previous ones, making the impacts worse over time (120). This might explain why even a mild head injury can lead to long-term adverse effects, especially if there have been previous untreated or asymptomatic traumas.
Emerging evidence suggests a critical window exists in the early hours to days post-injury for targeted metabolic intervention. Within minutes post-injury, the blood-brain barrier breaks down, triggering inflammation. By 1 hour, synapse density declines, disrupting neural communication. At 4 hours, inflammatory cytokines amplify tissue damage; and by 6 hours, mitochondrial dysfunction causes widespread energy failure. Immediate post-injury metabolic support may prevent extensive damage and accelerate recovery (20, 24).
The Vital Critical Window
Our understanding of the importance of the critical window of the early minutes/hours to days following concussion continues to expand. With a lack of robust studies in this arena to date, experts in the field advocate for “a logical application of science to the problem of concussion or a TBI (traumatic brain injury)”. In his 2020 seminal book Concussion Rescue, Dr. Kabran Chapek from Amen Clinics asks “what if we could immediately do something that increases the rate of recovery from traumatic brain injury, similar to how we routinely treat a sprained ankle with rest, ice, compression, and elevation?” Dr. Chapek advocates for a “TBI First Aid Kit”, including essential nutrients to support brain recovery, to be put to use within the first 24 hours post injury and continued for at least 7 days (72). His extensive clinical experience, and that of Amen Clinics, with TBI and concussion support his firm belief that targeted nutrient support immediately post injury is critical to support healing of the injured brain when the mechanisms of routine daily function fail (20).
Additional new studies also point to this potential window of opportunity to optimize metabolic support in order to better promote brain healing, showing detrimental impact within minutes post-injury.
-
- A 2021 review article titled “The synapse in traumatic brain injury” discusses the impact of traumatic brain injury (TBI) on synaptic structures. The authors highlight that TBI leads to rapid changes in synaptic proteins and a reduction in synapse density, particularly within the first 6 hours post-injury. For instance, studies have shown a significant decrease in synaptic density in the hippocampus as quickly as 1 hour after injury, with partial recovery observed over time (123).
-
- A 2013 study examining soldiers who suffered combat related head injury where they found that early use of the nutrient N-acetyl cysteine within 4 hours of injury led to resolution of symptoms in 86% as compared to only 42% in the placebo group (21). Rodent models of TBI likewise showed that early intervention within 24 hours of injury, with the nutrients acetyl-L-carnitine and citicoline respectively, demonstrated benefit in recovery (22).
Ever expanding research is clear that three critical brain systems falter early after concussion: energy production, management of inflammation, and elimination of toxins. It is well accepted that the brain is in a vulnerable condition immediately post-concussion with the first malfunction evident as an inability for the brain to use its most readily available energy source: glucose.
Following a brain injury, glucose transport into the cell often becomes impaired, creating an energy deficit during which the brain is unable to perform its essential functions of maintaining membrane potentials of nerve cells, quelling inflammation, and removing toxins. This leads to widespread dysfunction, most notably of astrocytes and synapses, the cells in the brain which utilize glucose to support surrounding neurons (123). After a traumatic brain injury (TBI), astrocytes, which usually help maintain a healthy brain environment, shift from a supportive mode to an inflammatory “reactive” mode termed “astrogliosis” (122). This reaction starts at the injury site and spreads to other areas of the brain. Near the injury, astrocytes cease some of their normal supportive functions and instead switch to producing more inflammatory substances. This shift from their usual role makes it more difficult for brain cells to survive and recover properly.
Additionally, these reactive astrocytes form a protective barrier, called a glial scar, to prevent harmful immune cells from spreading further into the brain. While this scar is helpful at first, it eventually blocks the repair of nerve connections, making it harder for the brain to regain normal function over time (122). This scaring ultimately hinders axonal reconnection and impairs long-term functional recovery, potentially affecting both local and global neural networks.
Astrocytes are also the only source of ketone body production in the brain, with ketones being an easily usable energy source for the brain when glucose is unavailable, such as during fasting (23). Thus when astrocytes no longer can utilize glucose nor produce ketones, an alternative power source, the brain now is in the precarious situation of “energy deficit”.
This knowledge opens up the opportunity to use exogenous (made outside the body) ketones as an accessible alternate power source. This current knowledge of brain science and the role of astrocytes supports Dr. Chapek’s focus on “a logical application of science to the problem of concussion or a TBI”, leading to the obvious question: How can we better support the recovery of failing brain mechanisms immediately post concussion (20)? Resolution of the immediate post injury energy crisis appears to be a crucial first step. Taking this a step further, how do we provide the brain with neuroprotective ingredients prior to high risk events such as sports or other high risk activities?
With a profound “energy deficit” the first site of malfunction, the brain then begins to falter in its routine daily activities. Inflammation, a normal response to the tissue damage of brain injury (12), is typically resolved by the body via anti-inflammatory cytokines and mobilization of key immune cells. With limited access to usable forms of energy, the mechanisms that guide the resolution of inflammation are impaired. Short-term consequences include the high likelihood of secondary injury with “neural priming” setting the stage for more severe damage with a second head injury (12). Over time, this may result in chronic neurodegeneration, dementia, Alzheimer’s, and encephalopathy (8,9,10). Application of current brain science suggests that targeted nutrients to quiet inflammation may be helpful in optimizing brain recovery post injury (20).
The ongoing “energy deficit” post injury then begins to impact another primary function of the brain: clearance of toxins. The timing here could not be worse, as immediately post concussion, damage to the blood brain barrier and cellular structures allow influx of toxins and inflammatory proteins, including damaged tissue, into the brain. Researchers at the Nedergaard Lab at the University of Rochester Medical Center, have delineated that post-TBI:
“Cell membranes and blood vessels rupture, spilling cytotoxic and inflammatory agents into the extracellular space and disrupting the blood supply, while blood-brain barrier breakdown leads to the influx of unfiltered blood born constituents, shutting down regular cellular metabolism.” (16).
The brain has a brilliant mechanism to remove toxins: the glymphatic system which, like the lymphatic system in the rest of the body, escorts toxins to the liver and then out via the intestinal tract or sweat. The glymphatic system is akin to your weekly garbage service, removing waste and toxins during sleep, essential for recovery (13,14). In the absence of an accessible energy source, glymphatics are just one of the multiple detoxification systems that deteriorate, allowing toxins and damaged tissue to remain in the brain, further augmenting the inflammatory response (15). Application of current brain science directs support for both cellular and glymphatic detoxification pathways in order to optimize recovery outcomes. This is supported by studies of the impact of key nutrients for detoxification in TBI recovery (24).
In summary, lacking robust clinical studies on acute interventions to date, the understanding of current neuroscience guides new support strategies for hindered brain metabolism post injury targeting energy, inflammation, and detoxification.
Current Recovery Strategies
The cornerstone of recovery recommendations has centered on “brain rest” for many decades.
According to the National Federation of State High School Associations (NFHS) and the Sports Medicine Advisory Committee (SMAC), the number one priority for concussion recovery is simple: rest (17). Their suggested guide for concussion management in sports claims that “The first step in recovering from a concussion is rest” (17) Sources like Johns Hopkins School of Medicine and the University of Michigan Health & Medicine publicly recommend and concur that full rest is paramount for concussion treatment and recovery. “In the first one to two days after suffering a concussion, near complete rest is important.” (18)
While traditional recovery recommendations have focused on ‘brain rest’ and reduction of neurostimulating activity until symptoms subsided, emerging literature now supports a return to exercise early in the course of mild TBI recovery as beneficial to the recovery timeline (25, 26, 27, 28).
Recent guidelines also emphasize the importance of cognitive and physical rehabilitation. The CDC submitted a report to Congress entitled Traumatic Brain Injury in the United States: Epidemiology and Rehabilitation, writing that TBI rehabilitation involves cognitive and physical therapies (19). Cognitive rehabilitation addresses cognitive deficits (learning, comprehension) and behavior, while physical rehabilitation focuses on improving mobility and ocular movement coordination.
These advancements in post concussion care, while leading to improved outcomes, continue to leave a gap in metabolic and anti-inflammatory support. Scientific advancements show that metabolic interventions during the early post-injury period can complement these approaches.
More research is clearly indicated, as the vulnerable window immediately following a concussion remains a potential opportunity where metabolic intervention could speed the brain in return to full function. The keys to a return to balance are the restoration of accessible energy for the brain, the resolution of inflammation, and the effective removal of toxins. Thus, a comprehensive treatment plan should integrate appropriate rest followed by a guided return to exercise early in the course of mild TBI recovery, cognitive and physical rehabilitation, as well as metabolic support.
The Consequences of Missed Interventions
Each year, about 1.4 million US citizens visit emergency rooms for traumatic brain injuries (29). Despite the most current protocols, about 50% of people with TBI will experience further decline in their daily lives or die within 5 years of their injury (30). Research published in the Journal of Head Trauma Rehabilitation demonstrated that at 10 years post-injury, the most common associated conditions developing after brain injury, in order, were back pain, depression, hypertension, anxiety, fractures, elevated cholesterol, sleep disorders, panic attacks, osteoarthritis, and diabetes (31). These disruptions can exacerbate symptoms and hinder recovery, illustrating the far-reaching consequences of brain injuries. These consequences involve multiple body systems including gastrointestinal, musculoskeletal, visual, cardiac, cognitive-behavioral, hormonal, sleep, as well as the integrity of the blood-brain barrier, a one cell thick layer of protection that allows in certain nutrients and excludes toxins from the brain.
Insights into Patient Experiences with Concussions
Frances Meredith, MD | This collection presents real-world accounts from patients who have personally experienced the failure of the current “gold standard” of care following concussions and traumatic brain injuries (TBIs). Each experience highlights the varied impacts of TBI, affecting cognitive function, physical health, and emotional well-being. By sharing these cases, we aim to provide a window into the complexity of the recovery journey with its profound impact on quality of life, as well as a look into different strategies and treatments utilized by these patients to set a course for return to a full and active life. These stories offer valuable perspectives which highlight evolving practices in concussion care.
CASE 1:
“A” presented to me, Dr. Meredith, as a busy 45-year-old mom with episodic flares of fatigue, brain fog, anxiety, joint discomfort, body pain, rapid heartbeat, and dizziness. She had previously been diagnosed with Hashimoto’s thyroiditis but hadn’t been informed it was autoimmune in nature. Her fatigue came in waves of “in-bed days,” often foreshadowed by a sense of feeling “dizzy but energetic” and knowing a crash was imminent. These episodes were often triggered by “doing too much” physically or emotionally or by reintroducing known reactive foods, such as sugar.
Her full Functional Medicine evaluation and treatment plan addressed underlying issues, including “leaky gut,” small intestinal bacterial overgrowth, chronic active Epstein-Barr virus (the mono virus), imbalanced hormonal levels including cortisol and Progesterone, and mold toxin exposure. Though these interventions led to a decrease in flare frequency, her symptoms continued to interfere with her quality of life. During a pivotal visit, we walked back through her history to answer the key question: “I was fine until ___.” In a moment of clarity, she identified a motor vehicle accident years earlier as the trigger for her health decline.
Her case illustrates the widespread effects of concussion, with gastrointestinal effects including increased intestinal permeability (“leaky gut”), and a tendency toward intestinal motility issues that promote small intestinal bacterial overgrowth (SIBO). Her episodic rapid heartbeat reflected cardiac dysfunction with imbalance in vagal nerve function and the autonomic nervous system underlying. Hormonal effects of her concussion were evident in her anxiety associated with abnormal cortisol levels. Finally cognitive-behavioral effects were evident in her brain fog, and her heightened anxiety reflecting both cerebellar dysfunction as well as vagal nerve imbalance.
CASE 2:
“B”, a 22-year-old 6’5” midfielder/center-back, was on track for MLS tryouts in 2023 when his path was altered. During training in the Northeast, he collided heads with another player while going up for a header and hit the ground unconscious. After regaining consciousness, the trainer checked him briefly and asked, “Do you feel okay? Do you want to go back in?” Running on adrenaline, “B” answered, “Yes!” but soon realized something was very wrong and took a knee on the field, marking the start of a long recovery process. Dr. Meredith has heard this story again and again. Research by Dr.Michael Collins at the University of Pittsburgh suggests each minute of play after a concussion can add 7–8 days to recovery.
Fortunately, “B” found a local concussion specialist who prescribed targeted visual exercises, Vitamin D, and creatine. Recovery, however, was slow and did not allow a return to play. Hoping to find someone to guide his healing to the point of being able to resume his professional soccer tryouts, “B” then sought care from a provider with a broader conceptual framework for assessing and treating concussion: Dr Andy Barlow in Tupelo, MI. Dr Barlow was well versed in Functional Medicine, allowing him an understanding of the total body effects of concussion. His extensive evaluation allowed a view into imbalances in multiple body systems including his vestibular (balance) system including the cerebellum and immune system with new antibodies to several body tissues reflecting the development of autoimmunity. In addition, Dr Barlow was well versed in evidence-based tools not used in traditional medicine for healing concussion such as Neurofeedback, red light therapy, Hyperbaric oxygen treatment, targeted elimination of inflammatory foods, and targeted supplement support.
Over weeks, “B” saw gradual improvements in physical activity and cognitive focus.
Inspired by the experiences of elite athletes, “B” explored psilocybin, a compound in “magic mushrooms,” within a ceremonial context. Two weeks after his session, his headaches finally subsided. Recent studies on psychedelics psilocybin and Ibogaine suggest they support TBI recovery through neuroplasticity, with research showing Ibogaine’s effects on neurotrophic factors and neuron growth. Although not widely available yet, these therapies offer promising advances for TBI treatments. A year after his TBI, even after significant healing, “B” continues to experience increased vulnerability to musculoskeletal injury not present prior, with a slow to heal hamstring injury after minimal provocation.
This case illustrates the often profound impact of concussion on multiple organ systems including the immune system, with head trauma triggering a cascade often leading to autoimmunity where the body begins to attack itself. This immune activation can open the door to future autoimmune or neurodegenerative disease such as Alzheimer’s or Parkinson’s disease.The effect of concussion on cerebellar function is evident in “B” as well, with its downstream effects on mood creating anxiety or “easy overwhelm”. His evaluation showed his cerebellar dysfunction to be profound.
The cerebellum has a little known key role in “gating” of information allowed forward in the brain to be processed by the frontal lobe. If too much information is allowed forward at one time, a scenario of “overload” and “overwhelm” is created, often leading to anxiety and insomnia. Musculoskeletal effects long after TBI are evident in his new tendency to injury not present prior.
CASE 3:
“B” has been a close friend of my (Dr. Meredith) son and family for many years. He reflects on the 7-8 concussions he’s endured since his early teens, beginning with soccer. At age 20, while teaching surfing, he suffered a concussion. Despite lingering symptoms, he returned to the water and was hit by a surfboard two weeks later. This brought debilitating migraines with severe light sensitivity, concentration difficulties, and intense reactions to sunlight and exercise—signs he chose to ignore. Complications arose with Dengue fever and later COVID, which, while his providers focused on these infections, led him to overlook the lingering impacts of his concussions. By early 2022, he resumed playing soccer, and after hitting his head snowboarding in 2023, his worst symptoms returned: fatigue, migraines after minimal cognitive exertion (even a 30-minute phone call), cognitive struggles, insomnia, brain fog, numbness, and a nervous system that “did not have an off switch.” Anxiety, a challenge since high school, escalated post-concussion with concurrent insomnia, poor appetite, and bowel issues. Seeking care from a concussion provider in Asheville, NC, testing revealed nervous system and eye movement dysfunction, which he described as his “eyes not keeping up” in busy environments like supermarkets or while driving, triggering migraines and dissociation. An intensive 14-day treatment focused on reaction times, eye movement exercises incorporating balance, and vagal nerve retraining. The program, continued at home, increased his tolerance for visual stimulation. “B” combined this neuro rehab with a supportive diet including bone broth, probiotic-rich foods, spore-based probiotics, L-glutamine, and an elimination diet. Fasting had a profound impact on his cognitive function, not a surprise with its effects on quieting inflammation and supercharging mitochondria, our cellular “energizer bunnies”. As he continues to heal, his ongoing challenge remains building his physical exertion tolerance. His message to others with concussion: “Prioritize the nervous system, especially sleep; eliminate substances that interfere with nervous system balance, and focus on the gut-brain connection, analyzing everything that goes into your body.”
His case illustrates the impact of concussion on the visual system with profound eye movement dysfunction improving with comprehensive rehabilitation. The cognitive, sleep, and anxiety challenges seen so often after concussion are evident in his case as well as the importance of the impact of concussion on the “gut-brain connection” with both leaky gut and small intestinal overgrowth, both common after head trauma.
CASE 4:
In February 2020, amidst COVID in her family, “E” began experiencing episodes of extreme exertional fatigue, sometimes confined to bed for 12-18 hours, particularly after elevating her heart rate with exercise. After her first COVID vaccine in March 2021, she awoke three days later with sudden, extreme fatigue that persisted to the point she could barely get out of bed. Initial evaluation revealed elevated Epstein-Barr virus antibodies (the virus underlying many cases of “mono”). Focusing on rest, she attempted more exercise June 2021 but felt she had “hit a wall,” with anxiety, insomnia, and the sense her nervous system was “buzzing” like “holding on to a low pulse electric fence.” She described brain fog, profound fatigue, “feeling like a zombie,” and heightened sensitivity to light and sound, making everyday interactions and activities unbearable. She could no longer be around noise and could no longer attend church. She found herself unable to calm her new extreme anxiety.
“E” sought Functional Medicine care with me (Dr. Meredith) in July 2021, where her treatment focused on rebalancing her stress axis, removing inflammatory triggers, and working with a cognitive behavioral therapist for sleep support. Feeling strongly that something was “off kilter” in her body, she began a trial of Effexor which compounded her anxiety and was discontinued. Recovery was gradual, with fatigue and headaches lingering even as her sleep and anxiety began to stabilize. Evaluation by Functional Neurologist Dr. Bianca Miret led to 12 hours of neuro-rehabilitation and 30 hours of hyperbaric oxygen therapy (HBOT), which significantly accelerated her recovery, especially impactful for her light and sound sensitivity, fatigue, and headaches. She continued managing inflammation with targeted supplements, an anti-inflammatory diet based on test-confirmed food sensitivities, and the elimination of seed oils.
After a COVID relapse in spring 2024, “E” experienced an increase in nervous system-related symptoms and began a COVID spike protein protocol to reduce lingering effects, which helped calm her nervous system. In 2024, she pursued further vagal nerve support and amygdala (part of the limbic system responsible for processing memory and emotions, especially fear) retraining through the somatic retraining within The Gupta Program, further accelerating her recovery. E now feels confident in her near-complete recovery and feels better prepared to manage future COVID-related flare-ups with anti-inflammatory tools.
Her case illustrates the profound effect of TBI, in this case a chemical TBI, on the central nervous system and the vagus nerve resulting in extreme anxiety as well as extreme reactivity to even minimal sensory stimuli. This added to the social isolation of her illness, only amplifying her anxiety.
CASE 5:
In June 2021, “H” received the mRNA COVID vaccine and immediately felt as if “my head was hit by a piece of wood,” followed by burning sensations throughout her body and brain, extreme disorientation and a sense of brain inflammation that kept her bedridden for five days. Symptoms began to improve but worsened again after a return to exercise three weeks later, triggering severe pressure at the base of her skull, new severe diarrhea, brain fog, disorientation, and head pressure that would shift to different areas. Severe visual and auditory issues followed, unpredictable, as well as pain she describes as “every nerve in the body just lit up” with pain and twitching. Worsening severe muscular weakness finally led to her first ER visit where she was sent home after being told she might have multiple sclerosis or rheumatoid arthritis.
New symptoms continued to surface unpredictably, including seizure-like episodes at the onset of sleep, episodic slurred speech, vomiting, severe acne, hair loss, impaired balance with a “rocking sensation” in the head, peripheral swelling, a sense of “buzzing in my tailbone”, soreness in joints, lack of coordination in her hand, and weight loss of 20 lbs over 3 weeks. Her severe vomiting led to another ER visit where life threatening low sodium levels were detected and her symptoms were attributed to these low levels.
Weeks later, anesthesia from a medical procedure re-triggered many of her original symptoms, leading to her “aha moment” realizing her symptoms were likely due to a reaction to chemicals. She shifted her focus toward rest and pursued recovery through vestibular physical therapy, visual rehab, neurofeedback with Dr. Robert Baric, and a Functional Medicine approach to inflammation, histamine sensitivity, and nutritional deficiencies. As of fall 2024, she feels “85% recovered,” still managing occasional brain fog. Her experience underscores the need for a supportive medical team and patience in recovery, emphasizing that concussion recovery is unique to each individual and often takes time. “H” wishes to spread awareness of the frustration and isolation caused by traumatic brain injuries, as well as the often-misunderstood symptoms.
The gastrointestinal effects of TBI, in this case of chemical origin, are very clear in this case with her post injury diarrhea likely a reflection of increased intestinal permeability or “leaky gut”. This case also illustrates the wide spread and confusing neural symptoms that are frequent post brain injury, as well as the immune effects evident in new joint pain.
Whole-Body Impact of Concussions: Effects Across Multiple Systems
We would like to take you on a tour of the many organ systems of the body impacted by concussion and TBI. It is clear that the effects of brain injury are body wide, not limited to the brain alone
Gastrointestinal
Numerous studies demonstrate a shift in gastrointestinal function following a traumatic brain injury (TBI). These shifts are felt to be both due to altered composition of the intestinal microbiome (the balance of bacteria, yeasts, viruses in the gut) as well as increased permeability of the intestinal barrier, also known as “leaky gut.” (34) A 2023 study from Loyola University showed that individuals after TBI have increased levels of the potentially pathogenic bacteria and decreased levels of bile acids in their feces and plasma (32).
These changes can foster maldigestion, intestinal dysfunction, and neuroinflammation, disrupting the balance of the bi-directional gut-brain axis, a “highway” connecting the intestinal tract and the brain. This has central nervous system (CNS) implications as at least 80% of serotonin is created in the intestinal tract; thus, neurotransmitter production is disrupted, impairing neuronal balance in the CNS (32).
Studies of the significance of intestinal permeability (leaky gut) following TBI in humans are growing in number. A 2024 study in the International Journal of Molecular Science studied military “breachers” exposed to repetitive low-level blasts, many with symptoms of mild traumatic brain injury. Biomarkers of intestinal permeability were assessed pre, post, and 24 hours after the blast and were found to be elevated (35).
Likewise, a 1998 study in the Journal of Trauma assessed gut permeability in TBI patients and detected impaired intestinal barrier function 4 days after injury; this breakdown of the intestinal barrier was correlated with disease severity and long-term prognosis (36).
There is general consensus based on multiple studies that intestinal permeability has a clear correlation with multiple forms of autoimmunity, with growing evidence of links to psychiatric disorders as well as neurodegenerative diseases, including Parkinson’s disease and Alzheimer’s. Thus, attention to repairing intestinal barrier integrity after TBI appears to be a vital component of a treatment plan aimed at prevention of these long-term chronic diseases.
Visual
Traumatic brain injury (TBI) can result in a variety of visual issues affecting different parts of the optic system (37). TBI, regardless of severity, is often associated with changes in all four categories of oculomotor behavior: convergence issues, fixations, smooth pursuits, and saccades (38). These impairments can manifest as dizziness with movement and car sickness due to malfunction in systems responsible for response inhibition, short-term spatial memory, motor-sequence programming, visuospatial processing, and visual attention (39). Literature trends suggest that impaired eye movement function correlates strongly with increased post-concussive symptom loads and difficulties in daily living activities (40). In addition the Pupillary Light Reflex is often negatively affected resulting in a myriad of symptoms from headaches, to eye fatigue and parasympathetic nervous system dysfunction.
Cardiac
“When concussions occur, the heart is not commonly thought of as a potentially damaged organ. But a surge in catecholamines from TBI can trigger arrhythmias and chronic cardiovascular disease. As a cardiologist, I advocate using Concusssion911 after TBI to help reduce systemic inflammation, boost antioxidant effects, and help stabilize the cell membrane in order to reduce potential cardiac complications.”
-Bill Passarelli, MD (Cardiology)
Traumatic brain injury causes a surge in catecholamines, which are hormones and neurotransmitters that regulate the fight-or-flight reaction, affecting cardiac cell receptors and leading to a systemic inflammatory response or “body on fire” (41, 42). This response creates cellular dysfunction, further damaging the brain and peripheral organs, including the heart. Among the complications of TBI, cardiac injury is a frequent occurrence, affecting approximately 25–35% of patients with TBI (42). The most common heart problems in people with TBI, occurring up to 5 to 10 times more often in individuals with TBI than in the general adult population (43) include a higher risk for developing chronic cardiovascular disease and irregular heart rhythms (42, 43). In addition, this parasympathetic dysfunction may negatively affect an individual’s Heart Rate Variability increasing ones mortality risks.
Musculoskeletal
“As a functional medicine professional, exercise physiologist and clinical care team member at the VA’s War Related Illness and Injury Study Center (WRIISC) I have a daily front row seat to the human cost of Traumatic Brain Injuries (TBI). Concussion911 would be my recommended first line intervention for anyone looking to optimize brain healing.”
-Elijah Sacra CEP, FMCHC (Clinical Exercise Physiologist, Functional Medicine Certified Health Coach)
A recent meta-analysis has highlighted a significant increase in the risk of subsequent injuries following an initial concussion. Specifically, individuals who have sustained a concussion have a 2.5 times greater risk of experiencing a subsequent musculoskeletal injury or a second concussion (44). This heightened risk is consistent across different levels of athletic competition, including professional, collegiate, and recreational athletes. Notably, the increased risk of injury remains present for up to 3 years following a concussion, well beyond the typical timeframe for concussion symptom resolution (44). TBI has also been linked to the abnormal development of bone tissue and increased callus formation (45) with disruption of endocrine factors and neuropeptides, which may adversely affect bone health and metabolism.
Blood-Brain Barrier
When a TBI is sustained, there is an immediate and direct impact on the blood-brain barrier, which may lead to detrimental long-term effects. The blood-brain barrier is a special additional screening mechanism that protects the brain from components in the blood entering into the protected area around the brain. This barrier dysfunction allows toxic materials usually excluded, such as bacterial lipopolysaccharide, environmental toxins, immune cells, and serum proteins, to enter the brain, worsening the inflammatory response already set in motion by tissue damage. The compromise of the blood-brain barrier further ignites the inflammatory cascade post injury (46, 47).
Cognitive-Behavioral
“As a Mental Health Counselor and Professor, I have witnessed some of the devastating consequences of concussions and TBIs, including anxiety, depression, panic attacks, sleep disturbances, cognitive impairments, elevated stress and suicidal ideation. Knowing that the early hours and days of a post-concussion injury represent a vital period for intervention to mitigate long-term damage, I recommend Concussion911 as a nutritional early intervention that optimizes recovery within this critical window post-injury. “
-Heloisa Portela, Ph.D., LPCS, NCC, ACS, RPT-S (Trauma focused therapist, professor)
Cognitive effects post-concussion are frequent, as seen in a study of 8,000 high school and college athletes which found the likelihood of developing Attention Deficit Hyperactivity Disorder (ADHD) or a Learning Disability (LD) were, respectively, 2.93 and 2.0 times higher in those with a history of concussion (48).
Individuals who have sustained a traumatic brain injury, regardless of the injury’s severity, are also more prone to developing chronic mood disorders and psychiatric conditions. These can include anger, depression, and anxiety, as well as post-traumatic stress disorder (PTSD). Depression and anxiety were noted in up to 30% of individuals post-injury (49). This is due not only in part to the psychological trauma associated with the incident, but also with neurotransmitter disruption and brain inflammation both adding insult to injury.
Studies have indicated that hippocampal damage occurs after TBI and can be a significant indicator of mood disorders in patients assessed post injury. Patients post TBI who developed mood disorders exhibited notably reduced hippocampal volumes (50).
Suicide, specifically the time-to-suicide, was thoroughly studied among 860,892 soldiers, including 108,785 who had experienced at least one documented TBI. The increase in mental health diagnoses from before to after TBI was significantly greater compared to those without a TBI history (51). Additionally, the time-to-suicide was 16.7% faster for soldiers with a TBI history than for those without.
Following a traumatic brain injury, anger and the ability to empathize are frequently reported concerns. From irritability to outbursts, up to one-third of patients report having negative behavioral symptoms following a concussion. Patients with TBI reported a decreased ability, compared to controls, to empathize emotionally, often associated with anger and low motivation (52).
The hippocampus, located deep within the brain, is also pivotal in laying down both short-term and long-term memory; thus it is not a surprise that TBI related damage results in deficits in working, declarative, and episodic memory. Individuals with moderate to severe head injuries were found to have considerably smaller hippocampal volumes compared to those with mild TBI. A comprehensive study on the cognitive effects of TBI demonstrated that individuals with a history of TBI performed worse on tasks involving executive function, attention, and memory compared to control groups (51).
These cognitive and emotional challenges also have a significant impact on caregivers. Caregivers of individuals with TBI often experience high levels of stress and burden, as they must manage not only the physical needs of the patient but also the psychological and behavioral changes (53). The demands of caregiving can lead to feelings of burnout, anxiety, and depression among caregivers. Research has shown that caregivers of TBI patients report lower quality of life and higher levels of psychological distress compared to caregivers of individuals with other chronic conditions (5).
Hormonal
TBI is associated with hormonal disruption on multiple levels (55).
A 2024 review in Brain and Spine highlighted several post-TBI neuroendocrine studies, identifying cortisol as the most frequently elevated hormone, peaking within 24 hours of injury (56). Heightened levels of cortisol can lead to a maladaptive inflammatory response, perpetuating dysfunction of both the blood-brain barrier and neuronal cells, leading to a plethora of negative symptoms (57). After the initial cortisol spike, long-term cortisol dysregulation amongst other adrenal hormones follows. Adrenal dysfunction is associated with fatigue, inadequate stress response, decreased memory, social issues, and mood or emotional disorders (58).
In addition to the disrupted adrenal axis, a broad range of pituitary hormone deficiencies are noted in one-third of adults and children post-TBI (58). As the pituitary gland produces hormones that stimulate testes and ovaries to produce estrogen, progesterone, and testosterone, it is not startling that studies confirm deficiencies in these reproductive hormones post-TBI (59). In a study of men with severe TBI, low testosterone levels were found in a high percentage (60).
Pituitary dysfunction has been found to be frequent following a traumatic brain injury and can include conditions like hypopituitarism, hypothyroidism, and hypogonadism. These conditions have a significant impact on executive function, concentration, problem-solving ability, memory, and speech (61).
Sleep
“Well thought out blend of supplements to support many aspects of brain healing.”
– Dr Singar Jagadeesan (Neurology, Sleep Medicine)
Fatigue, insomnia, attentional deficits, and impaired cognitive function are symptoms related to dysregulated sleep cycles following a traumatic brain injury (62). Affecting 30–70% of individuals post injury, disruption of natural circadian rhythms regulating the sleep/wake cycles can manifest as either insomnia or hypersomnia.
Traumatic brain injury patients with sleep disturbances have been found to have longer inpatient hospital stays, higher cost of rehabilitation, and higher rates of functional disability (63). Sleep disturbance post brain injury can also be impacted by pain elsewhere in the body, medications, and depression, much more common in persons with TBI than in the general population (49).
Progress and Gaps in Concussion Care
The landscape of TBI research is rapidly expanding, shedding light on the intricate nature of its diagnosis and understanding of its far-reaching systemic effects. There is a surge in the development of assessment protocols, as well as effective musculoskeletal, visual, and cognitive rehabilitation protocols. However, a significant gap exists in current multifaceted rehabilitation approaches in addressing acute prevention of the myriad metabolic impacts leading to far-reaching systemic consequences.
First, we will explore assessment tools. Current state of the art includes eye-movement tracking devices, now more standardized and better understood for their value in assessing the state of the visual and autonomic nervous system (66). As our understanding evolves, new methodologies are being explored to enhance assessment comprehensiveness. These include qEEG biomarkers, electrical signals recorded from the brain that can provide valuable insights into brain function and dysfunction. These qEEG biomarkers have shown promise in assessing the severity and prognosis of traumatic brain injury (65). Research indicates that quantitative EEG can identify, confirm, measure, and localize brain injury (65). These biomarkers help monitor cognitive function, detecting changes over time. Various blood tests are also being investigated as possible TBI biomarkers.
These advanced assessment methods offer a more detailed and holistic view of traumatic brain injuries. By using objective measurements, standardized procedures, and multi-faceted evaluation tools, clinicians can more accurately determine the severity and progression of concussions. This comprehensive approach helps understand the long-term effects of brain injuries and supports the development of more effective treatment and rehabilitation plans.
Current Treatment Recommendations
The latest evidence-based treatment recommendations are shaped by an evolving body of advanced research and clinical expertise. At the 6th International Conference on Concussion in Sport in Amsterdam in 2022, guidelines were updated with recommendations and protocols to standardize the assessment and management of concussions (67). These guidelines address various aspects of care, including initial evaluation, symptom monitoring, return-to-play criteria, and long-term management strategies. By providing a structured framework for assessment and treatment, the Amsterdam guidelines help ensure consistency and quality of care across different healthcare settings. One important area that is recommended is a thorough evaluation of the cervical spine by either a chiropractic physician or a specially trained physical therapist for assessment of ligament integrity and joint mobility.
The 2024 National Athletic Trainers Association (NATA) protocols for concussion management mark a significant step forward by promoting a more comprehensive approach to care (67). They now recommend early light exercise to enhance recovery, moving away from complete rest based on new evidence that physical activity aids brain healing. These protocols adopt a biopsychosocial model, integrating physical, psychological, and social factors, thus ensuring personalized treatment strategies that address mental health and social support needs. Educational initiatives have been strengthened, aiming to equip athletes, coaches, parents, and school administrators with thorough concussion knowledge to improve prevention, recognition, and management. The protocols emphasize multidimensional assessments, using cognitive tests, pre-competition qEEGs, and visual/vestibular screenings for tailored treatment plans.
Despite advancements in assessment, the treatment landscape for TBI remains relatively sparse, with only a few methods available to address the complex acute and chronic treatment needs of individuals affected by concussions. Efforts to develop a simplified, comprehensive approach are underway. One notable treatment approach is Vizstim, which promotes cognitive rehabilitation through a series of exercises designed to engage both the musculoskeletal and visual systems (68). By targeting these specific areas, Vizstim aims to improve cognitive efficiency over the long term. Other impactful treatment modalities with increasing support in the scientific literature include chiropractic spinal assessment and treatment, hyperbaric oxygen, Neurofeedback, evaluation by a chiropractic functional neurologist, as well as vagal nerve stimulation and retraining (70, 71, 72).
Multiple sources are now recommending targeted nutritional support for brain metabolism in the acute TBI phase, notably Dr. Kabran Chapek and other experts in the field from the Amen Clinic (69). Currently, however, there is no simple, easy to administer, systems-wide approach to address the magnitude of the brain’s unique metabolic demands during this vulnerable period. Advancements are needed in the realm of acute metabolic systems failures to bridge this gap and optimize outcomes.
A Comprehensive Solution: Introducing Concussion911
In response to the growing need for enhanced concussion care, a scientifically formulated nutritional solution appears to be the most viable option to support individuals immediately and in the acute phase after a suspected head injury. This innovative approach, Concussion911, emphasizes prompt intervention, optimization of brain energy production and utilization, and consumption of essential nutrients to foster recovery. By addressing these critical needs, our easy-to-use nutritional support combination aims to minimize the extent of damage, accelerate the healing process, and reduce the incidence of secondary complications.
Unlike conventional methods that primarily address symptom management, Concussion911 takes a proactive stance by leveraging the brain’s inherent mechanisms for healing and recovery. Supported by robust research and expert insights, this solution offers an effective and integrative method for managing concussions, bridging the gaps in current treatment. Through a carefully curated blend of nutrients, including vitamins, minerals, antioxidants, and neuroprotective compounds, we aim to support the multiple known system failure points with an optimized energy source, and compounds shown to both mitigate neuroinflammation and improve the removal of toxins from the brain and body.
Mitigating Neuroinflammation
Vitamin D
Vitamin D is not only a vitamin but functions as a hormone in many body processes (74, 76) . It has broad anti-inflammatory and neuroprotective effects, acting as an “immunomodulator” to dampen excessive immune responses (75). Vitamin D also helps to keep calcium in balance, which is important in TBI as excess calcium released in response to cellular damage fosters neuronal cell death (74, 75).
Omega-3
Omega-3 essential fatty acids, present in fish, eggs, algae, and nuts, have been shown to decrease the damage caused by brain injury (77, 78, 80, 81, 82). They are essential components of cell membranes, protecting cells from oxidative damage. The fatty acids EPA and DHA are both neuroprotective via multiple mechanisms, including increasing brain-derived neurotrophic factor (BDNF) (84). They are also essential in resolving inflammation via “resolvins” which act as proresolving mediators (83).
Curcumin
Curcumin, an active ingredient in turmeric, has been a well-known anti-inflammatory for centuries. In a mouse model of brain injury, curcumin was shown to decrease brain swelling following TBI and reduce interleukin-1, an inflammatory chemical released in the brain. It also decreased the activation of aquaporin-4, a water channel protein in cell membranes, including the blood-brain barrier and the glymphatic system. When activated, aquaporin-4 allows toxins to flow through to the usually well-protected brain (88). Curcumin exhibited beneficial immunomodulatory functions and protective capacities in multiple different TBI models (89,90, 91).
Pregnenolone
Pregnenolone is a neuroprotective and neuroactive inhibitory steroid that helps the nervous system to calm down. It has been found to be low in patients with a history of TBI. In a randomized study of 30 military veterans with mild TBI, pregnenolone has been shown to help with insomnia, irritability, and hypervigilance (all symptoms that overlap with post-concussive syndrome) (93).
Optimizing the Brain’s Energy Consumption
Ketones
Ketone bodies, produced in the liver as well as by brain astrocytes, are the most efficient fuel source for the brain (97, 98). Ketone bodies are naturally created in the fasting state. Studies in animals show that fasting after moderate TBI causes increase in ketones which are associated with neuroprotection, improved mitochondrial function, decreased oxidative stress, with maintenance of cognitive function (95). Exogenous ketones mitigate the “energetic crisis” evident within the first 24 hours of TBI when glucose transport falters, allowing the brain an alternative power source (23, 96). A 2024 review in Current Opinion in Clinical Nutrition and Metabolic Care highlights the opportunity for incorporating ketones in a comprehensive TBI management approach (94).
Acetyl-L carnitine
Acetyl-L-carnitine assists the mitochondria, our intracellular “Energizer bunnies”, in the production of energy. Acetyl-L-carnitine exerts neuroprotection via anti-inflammatory effects and as well as its effects on regulation of neuronal synaptic plasticity by counteracting post-trauma excitotoxicity (99, 100). In animal studies, it has been shown to reduce damage from TBI. In a 2010 study using a rat model of TBI, treatment with acetyl-L-carnitine during the first 24 hours after TBI improved behavioral outcomes and reduced brain damage within the first 7 days after injury in immature rats (20, 22). A 2023 rat TBI study also demonstrated that acetyl-L-carnitine treatment showed protective effects against those observed in the untreated rat TBI group (99).
Citicoline
Citicoline, or CDP-choline, is a component of human metabolism as an intermediate in the synthesis of phosphatidylcholine, the main phospholipid in cellular membranes, including neuronal and glial cells. It is also a factor in the balance of the acetylcholine neurotransmitter system necessary to carry messages from the brain to the rest of the body. Choline has shown promise in improving outcomes in patients post TBI (101, 102, 103, 104). A 2016 meta analysis of patients with head injury treated with citicoline within the first 24 hours showed increased independence in the follow up period (101). A 2018 retrospective matched pair analysis demonstrated reduced rates of intensive care unit (ICU) mortality, in-hospital mortality, 6‑month mortality, as well as of unfavorable outcomes (105).
While the largest human prospective trial of choline in TBI, the Citicoline Brain Injury Treatment Trial (COBRIT), failed to show a significant difference, there were methodologic issues with the study. COBRIT was a double-blind randomized and placebo-controlled trial of 1296 patients with mild, moderate or severe TBI (106). The inclusion of mild, moderate and severe TBI may have limited the power of the study to detect significant differences in these populations separately. In addition, sample size was not large enough to prove an optimal odds ratio, and compliance of only 44.4% of patients having taken more than 75% of the medication further limited the study design (106). Further study is clearly indicated.
Magnesium
Increasing evidence suggests that decline in Magnesium is a critical factor in acute brain injury, and that Magnesium supplementation can have beneficial impact on outcomes post brain injury (107, 108, 109, 110, 111, 121). A 2007 rat model exhibited magnesium chloride to be effective in facilitating cognitive recovery of function following brain injury (112). A followup study by the same author, also a rat model, demonstrated that magnesium treatment was successful at decreasing cognitive and motor deficits after TBI (113). Results of human studies have been mixed; as there are recently discovered variations of Magnesium chelates that can cross the blood brain barrier, including Magnesium threonate, more human studies are clearly needed.
Optimizing Detoxification Pathways
Several well studied essential nutrients for detoxification, both intracerebral and systemic, have been included in our nutrient supportive blend.
N-acetyl Cysteine (NAC)
N-acetyl Cysteine (NAC), as a precursor to Glutathione, is a powerful antioxidant and supports removal of harmful toxins and chemicals from body and brain. It is critical in the detoxification of reactive oxygen and nitrogen species which create cellular damage (114). A 2013 randomized controlled trial involving 81 military personnel with artillery-blast head injuries demonstrated that those treated with oral NAC within 24 hours of injury experienced a significant improvement in symptom resolution compared to the control group. The study reported that 86% of the NAC group had complete symptom resolution compared to 42% in the placebo group (85). A 2013 study demonstrated that impaired glutathione synthesis is linked to increased risk for neurologic disease (86). Topical glutathione reduced cell death in an experimental model of TBI in mice (87).
Vitamin C
Further brain injury, known as “secondary injury,” is felt to be due to several mechanisms, including oxidative stress. Vitamin C, a powerful antioxidant, becomes quickly depleted during times of critical illness, allowing the damage cascade to continue. Vitamin C has been shown to improve outcomes in patients with acute TBI (24, 92), mitigating increased reactive oxygen species and free radicals associated with tissue damage. It is an essential nutrient in system wide detoxification processes (115).
Additional Support
Creatine
Creatine has shown promise for attenuating symptoms of concussion, mild TBI and depression (116, 117, 118). A 2023 review of 15 articles supports the benefit of creatine in patients post TBI (118). Of note, results are only clinically significant after a month of supplementation. Creatine showed efficacy as a neuroprotective agent in battling the chronic manifestations which lead to oxidative stress and cognitive function post brain injury (118).
Conclusion
A Whole-Body Approach to Recovery
Kali Regan | Throughout our journeys, we have witnessed firsthand the profound, life-altering consequences of traumatic brain injuries (TBIs). Though our paths to this understanding were different, as most are, they led us to a shared realization: TBIs are far more than just a “head injury.” The effects ripple throughout the body, often unnoticed or misunderstood, with the damage accumulating over time.
I still vividly remember my first concussion. It seemed so simple then—a blow to the head, some rest, and back to the game. But as time went on, the hits kept coming. Over six years, I sustained more than seven concussions, each one stacking on top of the last. We were told to rest, to avoid risk, but no one warned us about the subtle and far-reaching metabolic consequences of these injuries. I didn’t fully grasp the weight of the damage until it became too much to ignore.
After my final concussion, I woke up on the field, disoriented, bloody and bruised, unaware that my brain was in crisis. I ignored the signs—rushing back to the game within a few days, oblivious to the long-term consequences of my actions. Like many athletes, I believed in the myth that recovery was just a matter of taking a few days off. But I quickly learned the hard way that this couldn’t have been further from the truth. The damage was cumulative, and each hit made the next more dangerous.
IN SUMMARY
The countless stories and evidence showing the need for a new approach to healing motivated us— that’s why we created Concussion911. Our experiences have shown us that true recovery takes more than just rest. It requires a proactive, whole-body approach. It requires understanding the metabolic demands of the brain in the critical moments following injury, supporting it through nutrients that promote healing at the cellular level.
Despite the significant strides made in concussion research, there remains a critical gap in our understanding of the body’s needs in the first hours following injury. While protocols have improved, the science is still evolving, and much work remains to be done. We recognized this gap and are eager to promote Concussion911 as a response—targeted, effective support during the crucial acute phase of recovery.
Our formulation doesn’t just address symptoms; it supports the metabolic pathways that allow for true healing, bridging the gap between what’s currently accessible and what’s undoubtedly needed in the aftermath of a concussion. It’s more than just an intervention—it’s a lifeline.
We are committed to ensuring that future generations don’t have to face the same uncertainties and long-term health risks we, our families, and our patients did. The impact of concussion is vast, and we are only beginning to understand its true extent. But with a holistic, informed approach, we can help shape a future where brain health is truly prioritized.
Concussion911 is more than a product—it’s a movement. A movement towards understanding, healing, and proactively protecting our brains, not just in the immediate aftermath of a hit, but for a lifetime.
CONTACT
Kali Regan, info@myconcussiondr.com
Safety Considerations
Contraindications to Concussion911 are pregnancy, breast feeding, and active cancer treatment. If you are taking blood thinners consult with your doctor before beginning Concussion911. Do not take additional Vit D, creatine, or fish oil while using Concussion911. Consult with your doctor if you are taking pharmaceutical medications in order to rule out potential interactions.
If you feel your concussion is due to chemical induced brain injury, consult your TBI informed provider before starting this supplement and consider starting at tiny doses due to increased potential for unpredictable reactivity. As with all supplements and medications, there is individual variability in tolerance.
References
-
- Traumatic brain injury. American Brain Foundation. (2023, May 26). https://www.americanbrainfoundation.org/diseases/traumatic-brain-injury/
-
- DePadilla, L., Miller, G. F., Jones, S. E., Peterson, A. B., & Breiding, M. J. (2018). Self-reported concussions from playing a sport or being physically active among high school students — United States, 2017. MMWR. Morbidity and Mortality Weekly Report, 67(24), 682–685. https://doi.org/10.15585/mmwr.mm6724a3
-
- Hiploylee, C., Dufort, P. A., Davis, H. S., Wennberg, R. A., Tartaglia, M. C., Mikulis, D., Hazrati, L.-N., & Tator, C. H. (2017). Longitudinal Study of Postconcussion Syndrome: Not everyone recovers. Journal of Neurotrauma, 34(8), 1511–1523. https://doi.org/10.1089/neu.2016.4677
-
- Mucha, A., & Trbovich, A. (2019). Considerations for diagnosis and management of concussion. Journal of Orthopaedic & Sports Physical Therapy, 49(11), 787–798. https://doi.org/10.2519/jospt.2019.8855
-
- Lozano, D., Schimmel, S., & Acosta, S. (2017). Neuroinflammation in traumatic brain injury: A chronic response to an acute injury. Brain Circulation, 3(3), 135. https://doi.org/10.4103/bc.bc_18_17
-
- Manley, G., Gardner, A. J., Schneider, K. J., Guskiewicz, K. M., Bailes, J., Cantu, R. C., Castellani, R. J., Turner, M., Jordan, B. D., Randolph, C., Dvořák, J., Hayden, K. A., Tator, C. H., McCrory, P., & Iverson, G. L. (2017). A systematic review of potential long-term effects of sport-related concussion. British Journal of Sports Medicine, 51(12), 969–977. https://doi.org/10.1136/bjsports-2017-097791
-
- Lozano, D., Schimmel, S., & Acosta, S. (2017). Neuroinflammation in traumatic brain injury: A chronic response to an acute injury. Brain Circulation, 3(3), 135. https://doi.org/10.4103/bc.bc_18_17
-
- Acosta, S. A., Tajiri, N., de la Pena, I., Bastawrous, M., Sanberg, P. R., Kaneko, Y., & Borlongan, C. V. (2015). Alpha‐synuclein as a pathological link between chronic traumatic brain injury and parkinson’s disease. Journal of Cellular Physiology, 230(5), 1024–1032. https://doi.org/10.1002/jcp.24830
-
- Uryu, K., Chen, X.-H., Martinez, D., Browne, K. D., Johnson, V. E., Graham, D. I., Lee, V. M.-Y., Trojanowski, J. Q., & Smith, D. H. (2007). Multiple proteins implicated in neurodegenerative diseases accumulate in axons after brain trauma in humans. Experimental Neurology, 208(2), 185–192. https://doi.org/10.1016/j.expneurol.2007.06.018
-
- Tajiri, N., Kellogg, S. L., Shimizu, T., Arendash, G. W., & Borlongan, C. V. (2013). Traumatic brain injury precipitates cognitive impairment and extracellular AΒ aggregation in alzheimer’s disease transgenic mice. PLoS ONE, 8(11). https://doi.org/10.1371/journal.pone.0078851
-
- Erta, M., Quintana, A., & Hidalgo, J. (2012). Interleukin-6, a major cytokine in the central nervous system. International Journal of Biological Sciences, 8(9), 1254–1266. https://doi.org/10.7150/ijbs.4679
-
- Kalra, S., Malik, R., Singh, G., Bhatia, S., Al-Harrasi, A., Mohan, S., Albratty, M., Albarrati, A., & Tambuwala, M. M. (2022). Pathogenesis and management of Traumatic Brain Injury (TBI): Role of neuroinflammation and anti-inflammatory drugs. Inflammopharmacology, 30(4), 1153–1166. https://doi.org/10.1007/s10787-022-01017-8
-
- Anthony L. Komaroff, M. (2021, July 1). Are toxins flushed out of the brain during sleep?. Harvard Health. https://www.health.harvard.edu/mind-and-mood/are-toxins-flushed-out-of-the-brain-during-sleep#:~:text=The%20waste%20management%20system%20%28called%20the%20glymphatic%20system%29,the%20blood.%20This%20occurs%20primarily%20during%20deep%20sleep.
-
- Ferrara, M., Bertozzi, G., Volonnino, G., Di Fazio, N., Frati, P., Cipolloni, L., La Russa, R., & Fineschi, V. (2022). Glymphatic system a window on TBI Pathophysiology: A systematic review. International Journal of Molecular Sciences, 23(16), 9138. https://doi.org/10.3390/ijms23169138
-
- Reddy, O. C., & van der Werf, Y. D. (2020). The sleeping brain: Harnessing the power of the Glymphatic system through lifestyle choices. Brain Sciences, 10(11), 868. https://doi.org/10.3390/brainsci10110868
-
- Nedergaard, M. (n.d.). Glial responses to traumatic brain injury, stroke, the role of gliosis. University of Rochester Medical Center. https://www.urmc.rochester.edu/labs/nedergaard/projects/glial-responses-to-traumatic-brain-injury-stroke.aspx
-
- Suggested-guidelines-for-management-of-concussion-in-sports (2002). https://www.nfhs.org/media/1020401/suggested-guidelines-for-management-of-concussion-in-sports-april-2019-final.pdf
-
- Concussion treatment and recovery. Concussion Treatment and Recovery | University of Michigan Health. (n.d.). https://www.uofmhealth.org/conditions-treatments/brain-neurological-conditions/concussion-treatment-and-recovery
-
- Centers for Disease Control and Prevention. (2015). Report to Congress on Traumatic Brain Injury in the United States: Epidemiology and Rehabilitation. National Center for Injury Prevention and Control; Division of Unintentional Injury Prevention. Atlanta, GA.
-
- Chapek, K., & Amen, D. G. (2020). Concussion rescue: A comprehensive program to heal traumatic brain injury. Citadel Press.
-
- Hoffer, M. E., Balaban, C., Slade, M. D., Tsao, J. W., & Hoffer, B. (2013). Amelioration of acute sequelae of blast induced mild traumatic brain injury by N-acetyl cysteine: A double-blind, placebo controlled study. PLoS ONE, 8(1). https://doi.org/10.1371/journal.pone.0054163
-
- Scafidi, S., Racz, J., Hazelton, J., McKenna, M. C., & Fiskum, G. (2010). Neuroprotection by acetyl-l-carnitine after traumatic injury to the immature rat brain. Developmental Neuroscience, 32(5–6), 480–487. https://doi.org/10.1159/000323178
-
- Zhang, Y., Qi, Y., Gao, Y., Chen, W., Zhou, T., Zang, Y., & Li, J. (2023). Astrocyte metabolism and signaling pathways in the CNS. Frontiers in Neuroscience, 17. https://doi.org/10.3389/fnins.2023.1217451
-
- Shen, Q., Hiebert, J. B., Hartwell, J., Thimmesch, A. R., & Pierce, J. D. (2016). Systematic review of traumatic brain injury and the impact of antioxidant therapy on clinical outcomes. Worldviews on Evidence-Based Nursing, 13(5), 380–389. https://doi.org/10.1111/wvn.12167
-
- Chin, L. M., Keyser, R. E., Dsurney, J., & Chan, L. (2015). Improved cognitive performance following aerobic exercise training in people with traumatic brain injury. Archives of Physical Medicine and Rehabilitation, 96(4), 754–759. https://doi.org/10.1016/j.apmr.2014.11.009
-
- Leddy, J. J., Wilber, C. G., & Willer, B. S. (2018). Active recovery from concussion. Current Opinion in Neurology, 31(6), 681–686. https://doi.org/10.1097/wco.0000000000000611
-
- Romanov, R., Mesarič, L., Perić, D., Vešligaj Damiš, J., & Petrova Filišič, Y. (2021). The effects of adapted physical exercise during rehabilitation in patients with traumatic brain injury. Turkish Journal of Physical Medicine and Rehabilitation, 67(4), 482–489. https://doi.org/10.5606/tftrd.2021.6145
-
- Leddy, J. J., Haider, M. N., Ellis, M., & Willer, B. S. (2018b). Exercise is medicine for concussion. Current Sports Medicine Reports, 17(8), 262–270. https://doi.org/10.1249/jsr.0000000000000505
-
- Lozano, D., Schimmel, S., & Acosta, S. (2017a). Neuroinflammation in traumatic brain injury: A chronic response to an acute injury. Brain Circulation, 3(3), 135. https://doi.org/10.4103/bc.bc_18_17
-
- Centers for Disease Control and Prevention. (n.d.). About mild TBI and concussion. Centers for Disease Control and Prevention. https://www.cdc.gov/traumatic-brain-injury/about/index.html
-
- Lozano, D., Schimmel, S., & Acosta, S. (2017a). Neuroinflammation in traumatic brain injury: A chronic response to an acute injury. Brain Circulation, 3(3), 135. https://doi.org/10.4103/bc.bc_18_17
-
- Cannon, A. R., Anderson, L. J., Galicia, K., Murray, M. G., Kamran, A. S., Li, X., Gonzalez, R. P., & Choudhry, M. A. (2023). Traumatic brain injury induced inflammation and GI motility dysfunction. Shock. https://doi.org/10.1097/shk.0000000000002082
-
- Hanscom, M., Loane, D. J., & Shea-Donohue, T. (2021). Brain-gut axis dysfunction in the pathogenesis of Traumatic Brain Injury. Journal of Clinical Investigation, 131(12). https://doi.org/10.1172/jci143777
-
- Hang, C.-H. (2003a). Alterations of intestinal mucosa structure and barrier function following traumatic brain injury in rats. World Journal of Gastroenterology, 9(12), 2776. https://doi.org/10.3748/wjg.v9.i12.2776
-
- X, V. A. R. C. (2024, April 5). Blast exposure linked to intestinal problems. Medical Xpress – medical research advances and health news. https://medicalxpress.com/news/2024-04-blast-exposure-linked-intestinal-problems.html#:~:text=Researchers%20found%20biomarkers%20of%20intestinal%20permeability%20and%20signs,hours%20after%20blast%20exposure%2C%20suggesting%20a%20direct%20connection.
-
- Faries, P. L., Simon, R. J., Martella, A. T., Lee, M. J., & Machiedo, G. W. (1998). Intestinal permeability correlates with severity of injury in trauma patients. The Journal of Trauma: Injury, Infection, and Critical Care, 44(6), 1031–1036. https://doi.org/10.1097/00005373-199806000-00016
-
- Richman, E. A. (2016, May 4). Traumatic brain injury and visual disorders: What every ophthalmologist should know. American Academy of Ophthalmology. https://www.aao.org/eyenet/article/traumatic-brain-injury-visual-disorders-what-every-2
-
- Hunfalvay, M., Roberts, C.-M., Murray, N., Tyagi, A., Kelly, H., & Bolte, T. (2019). Horizontal and vertical self-paced saccades as a diagnostic marker of traumatic brain injury. Concussion, 4(1). https://doi.org/10.2217/cnc-2019-0001
-
- Heitger, M. H., Jones, R. D., Macleod, A. D., Snell, D. L., Frampton, C. M., & Anderson, T. J. (2009b). Impaired eye movements in post-concussion syndrome indicate suboptimal brain function beyond the influence of depression, malingering or intellectual ability. Brain, 132(10), 2850–2870. https://doi.org/10.1093/brain/awp181
-
- Armstrong, R. A. (2018). Visual problems associated with traumatic brain injury. Clinical and Experimental Optometry, 101(6), 716–726. https://doi.org/10.1111/cxo.12670
-
- Christensen, T. (2020, November 11). Traumatic brain injury, PTSD boost heart attack risk in veterans. www.heart.org. https://www.heart.org/en/news/2020/11/11/traumatic-brain-injury-ptsd-boost-heart-attack-risk-in-veterans
-
- Coppalini, G., Salvagno, M., Peluso, L. et al. Cardiac Injury After Traumatic Brain Injury: Clinical Consequences and Management. Neurocrit Care 40, 477–485 (2024). https://doi.org/10.1007/s12028-023-01777-3
-
- Izzy, S., Grashow, R., Radmanesh, F., Chen, P., Taylor, H., Formisano, R., Wilson, F., Wasfy, M., Baggish, A., & Zafonte, R. (2023). Long-term risk of cardiovascular disease after traumatic brain injury: Screening and prevention. The Lancet Neurology, 22(10), 959–970. https://doi.org/10.1016/s1474-4422(23)00241-7
-
- Smulligan, K. L., Wilson, J. C., & Howell, D. R. (2022). Increased risk of musculoskeletal injuries after concussion. Operative Techniques in Sports Medicine, 30(1), 150896. https://doi.org/10.1016/j.otsm.2022.150896
-
- Bajwa, N. M., Kesavan, C., & Mohan, S. (2018). Long-term consequences of traumatic brain injury in Bone metabolism. Frontiers in Neurology, 9. https://doi.org/10.3389/fneur.2018.00115
-
- Cash, A., & Theus, M. H. (2020). Mechanisms of blood–brain barrier dysfunction in traumatic brain injury. International Journal of Molecular Sciences, 21(9), 3344. https://doi.org/10.3390/ijms21093344
-
- Xiong, Y., Mahmood, A., & Chopp, M. (2018). Current understanding of neuroinflammation after traumatic brain injury and cell-based therapeutic opportunities. Chinese Journal of Traumatology, 21(3), 137–151. https://doi.org/10.1016/j.cjtee.2018.02.003
-
- Nelson, L. D., Guskiewicz, K. M., Marshall, S. W., Hammeke, T., Barr, W., Randolph, C., & McCrea, M. A. (2016). Multiple self-reported concussions are more prevalent in athletes with ADHD and learning disability. Clinical Journal of Sport Medicine, 26(2), 120–127. https://doi.org/10.1097/jsm.0000000000000207
-
- Jorge, R. E., Robinson, R. G., Moser, D., Tateno, A., Crespo-Facorro, B., & Arndt, S. (2004). Major depression following traumatic brain injury. Archives of General Psychiatry, 61(1), 42. https://doi.org/10.1001/archpsyc.61.1.42
-
- Jorge, R. E., Acion, L., Starkstein, S. E., & Magnotta, V. (2007). Hippocampal volume and mood disorders after traumatic brain injury. Biological Psychiatry, 62(4), 332–338. https://doi.org/10.1016/j.biopsych.2006.07.024
-
- Brenner, L. A., Forster, J. E., Gradus, J. L., Hostetter, T. A., Hoffmire, C. A., Walsh, C. G., Larson, M. J., Stearns-Yoder, K. A., & Adams, R. S. (2023). Associations of military-related traumatic brain injury with new-onset mental health conditions and suicide risk. JAMA Network Open, 6(7). https://doi.org/10.1001/jamanetworkopen.2023.26296
-
- de Sousa, A., McDonald, S., & Rushby, J. (2012). Changes in emotional empathy, affective responsivity, and behavior following severe traumatic brain injury. Journal of Clinical and Experimental Neuropsychology, 34(6), 606–623. https://doi.org/10.1080/13803395.2012.667067
-
- Verhaeghe, S., Defloor, T., & Grypdonck, M. (2005). Stress and coping among families of patients with traumatic brain injury: A review of the literature. Journal of Clinical Nursing, 14(8), 1004–1012. https://doi.org/10.1111/j.1365-2702.2005.01126.x
-
- Kreutzer, J. S., Rapport, L. J., Marwitz, J. H., Harrison-Felix, C., Hart, T., Glenn, M., & Hammond, F. (2009). Caregivers’ well-being after traumatic brain injury: A multicenter prospective investigation. Archives of Physical Medicine and Rehabilitation, 90(6), 939–946. https://doi.org/10.1016/j.apmr.2009.01.010
-
- Bollerslev, J., Klibanski, A., & Tritos, N. (2013). Traumatic brain injury: effects on the endocrine system. The Journal of clinical endocrinology and metabolism, 98(3), 27A–8A. https://doi.org/10.1210/jcem.98.3.zeg27a
-
- Magyar-Sumegi, Z. D., Stankovics, L., Lendvai-Emmert, D., Czigler, A., Hegedus, E., Csendes, M., Toth, L., Ungvari, Z., Buki, A., & Toth, P. (2024). Acute neuroendocrine changes after traumatic brain injury. Brain & spine, 4, 102830. https://doi.org/10.1016/j.bas.2024.102830
-
- Barton, D. J., Kumar, R. G., Schuster, A. A., Juengst, S. B., Oh, B.-M., & Wagner, A. K. (2021). Acute cortisol profile associations with cognitive impairment after severe traumatic brain injury. Neurorehabilitation and Neural Repair, 35(12), 1088–1099. https://doi.org/10.1177/15459683211048771
-
- Mahajan, C., Prabhakar, H. & Bilotta, F. Endocrine Dysfunction After Traumatic Brain Injury: An Ignored Clinical Syndrome?. Neurocrit Care 39, 714–723 (2023). https://doi.org/10.1007/s12028-022-01672-3
-
- Lopez-Rodriguez, A. B., Acaz-Fonseca, E., Spezzano, R., Giatti, S., Caruso, D., Viveros, M.-P., Melcangi, R. C., & Garcia-Segura, L. M. (2016). Profiling neuroactive steroid levels after traumatic brain injury in male mice. Endocrinology, 157(10), 3983–3993. https://doi.org/10.1210/en.2016-1316
-
- Hohl, A., Zanela, F. A., Ghisi, G., Ronsoni, M. F., Diaz, A. P., Schwarzbold, M. L., Dafre, A. L., Reddi, B., Lin, K., Pizzol, F. D., & Walz, R. (2018). Luteinizing hormone and testosterone levels during acute phase of severe traumatic brain injury: Prognostic implications for adult male patients. Frontiers in Endocrinology, 9. https://doi.org/10.3389/fendo.2018.00029
-
- Gilis-Januszewska, A., Kluczyński, Ł., & Hubalewska-Dydejczyk, A. (2020b). Traumatic brain injuries induced pituitary dysfunction: A call for algorithms. Endocrine Connections, 9(5). https://doi.org/10.1530/ec-20-0117
-
- Gottesman, R. F., Lutsey, P. L., Benveniste, H., Brown, D. L., Full, K. M., Lee, J.-M., Osorio, R. S., Pase, M. P., Redeker, N. S., Redline, S., & Spira, A. P. (2024). Impact of sleep disorders and disturbed sleep on Brain Health: A Scientific Statement from the American Heart Association. Stroke, 55(3). https://doi.org/10.1161/str.0000000000000453
-
- Lim, M. M., Elkind, J., Xiong, G., Galante, R., Zhu, J., Zhang, L., Lian, J., Rodin, J., Kuzma, N. N., Pack, A. I., & Cohen, A. S. (2013). Dietary therapy mitigates persistent wake deficits caused by mild traumatic brain injury. Science Translational Medicine, 5(215). https://doi.org/10.1126/scitranslmed.3007092
-
- Kocik, V. I., Dengler, B. A., Rizzo, J. A., MA Moran, M., Willis, A. M., April, M. D., & Schauer, S. G. (2023). A narrative review of existing and developing biomarkers in acute traumatic brain injury for potential military deployed use. Military Medicine, 189(5–6). https://doi.org/10.1093/milmed/usad433
-
- Nishimura, K., Cordeiro, J. G., Ahmed, A. I., Yokobori, S., & Gajavelli, S. (2022). Advances in traumatic brain injury biomarkers. Cureus. https://doi.org/10.7759/cureus.23804
-
- Alhilo, T., & Al-Sakaa, A. (2024). Eye Tracking Review: Importance, tools, and applications. Lecture Notes in Networks and Systems, 383–394. https://doi.org/10.1007/978-3-031-56728-5_32
-
- Broglio, S. P., Register-Mihalik, J. K., Guskiewicz, K. M., Leddy, J. J., Merriman, A., & Valovich McLeod, T. C. (2024). National Athletic Trainers’ Association bridge statement: Management of sport-related concussion. Journal of Athletic Training, 59(3), 225–242. https://doi.org/10.4085/1062-6050-0046.22
-
- VIZSTIM: Visual & Vestibular Home Exercise Kits. VIZSTIM LLC. (n.d.). https://vizstim.com/
-
- Carneiro, L., & Pellerin, L. (2022). Nutritional impact on metabolic homeostasis and Brain Health. Frontiers in Neuroscience, 15. https://doi.org/10.3389/fnins.2021.767405
-
- Bennett, M. (2018). Evidence brief: Hyperbaric oxygen therapy (HBOT) for traumatic brain injury and/or post-traumatic stress disorder. Diving and Hyperbaric Medicine Journal, 48(2), 115–115. https://doi.org/10.28920/dhm48.2.115
-
- Surmeli, T., Eralp, E., Mustafazade, I., Kos, H., Özer, G. E., & Surmeli, O. H. (2015). Quantitative EEG neurometric analysis–guided neurofeedback treatment in dementia. Clinical EEG and Neuroscience, 47(2), 118–133. https://doi.org/10.1177/1550059415590750
-
- Shi, C., Flanagan, S. R., & Samadani, U. (2013). Vagus nerve stimulation to augment recovery from severe traumatic brain injury impeding consciousness: A prospective pilot clinical trial. Neurological Research, 35(3), 263–276. https://doi.org/10.1179/1743132813y.0000000167
-
- Kabran Chapek, Nd: Seattle Metro Area. Kabran Chapek, ND | Amen Clinics Amen Clinics. (n.d.). https://www.amenclinics.com/team/kabran-chapek-nd/
-
- Sassi, F., Tamone, C., & D’Amelio, P. (2018). Vitamin D: Nutrient, hormone, and immunomodulator. Nutrients, 10(11), 1656. https://doi.org/10.3390/nu10111656
-
- Thau-Zuchman, O., Gomes, R. N., Dyall, S. C., Davies, M., Priestley, J. V., Groenendijk, M., De Wilde, M. C., Tremoleda, J. L., & Michael-Titus, A. T. (2019). Brain phospholipid precursors administered post-injury reduce tissue damage and improve neurological outcome in experimental traumatic brain injury. Journal of Neurotrauma, 36(1), 25–42. https://doi.org/10.1089/neu.2017.5579
-
- Scrimgeour, A. G., Condlin, M. L., Loban, A., & DeMar, J. C. (2021). Omega-3 fatty acids and vitamin D decrease plasma T-Tau, GFAP, and Uch-L1 in experimental traumatic brain injury. Frontiers in Nutrition, 8. https://doi.org/10.3389/fnut.2021.685220
-
- Thau-Zuchman, O., Gomes, R. N., Dyall, S. C., Davies, M., Priestley, J. V., Groenendijk, M., De Wilde, M. C., Tremoleda, J. L., & Michael-Titus, A. T. (2019). Brain Phospholipid Precursors Administered Post-Injury Reduce Tissue Damage and Improve Neurological Outcome in Experimental Traumatic Brain Injury. Journal of neurotrauma, 36(1), 25–42. https://doi.org/10.1089/neu.2017.5579
-
- Wu, Y., Zhang, J., Feng, X., & Jiao, W. (2023). Omega-3 polyunsaturated fatty acids alleviate early brain injury after traumatic brain injury by inhibiting neuroinflammation and necroptosis. Translational neuroscience, 14(1), 20220277. https://doi.org/10.1515/tnsci-2022-0277
-
- Michael-Titus, A. T. (2007). Omega-3 fatty acids and neurological injury. Prostaglandins, Leukotrienes and Essential Fatty Acids, 77(5–6), 295–300. https://doi.org/10.1016/j.plefa.2007.10.021
-
- Finnegan, E., Daly, E., Pearce, A. J., & Ryan, L. (2022). Nutritional interventions to support acute mtbi recovery. Frontiers in Nutrition, 9. https://doi.org/10.3389/fnut.2022.977728
-
- Patch, C. S., Hill-Yardin, E. L., Lewis, M., Ryan, L., Daly, E., & Pearce, A. J. (2021). The more, the better: High-dose omega-3 fatty acids improve behavioural and molecular outcomes in preclinical models in mild brain injury. Current Neurology and Neuroscience Reports, 21(9). https://doi.org/10.1007/s11910-021-01132-z
-
- Scrimgeour, A. G., Condlin, M. L., Loban, A., & DeMar, J. C. (2021a). Omega-3 fatty acids and vitamin D decrease plasma T-Tau, GFAP, and Uch-L1 in experimental traumatic brain injury. Frontiers in Nutrition, 8. https://doi.org/10.3389/fnut.2021.685220
-
- Norling, L. V., & Serhan, C. N. (2010). Profiling in resolving inflammatory exudates identifies novel anti‐inflammatory and Pro‐resolving mediators and signals for termination. Journal of Internal Medicine, 268(1), 15–24. https://doi.org/10.1111/j.1365-2796.2010.02235.x
-
- Ziaei, S., Mohammadi, S., Hasani, M., Morvaridi, M., Belančić, A., Daneshzad, E., Saleh, S. A., Adly, H. M., & Heshmati, J. (2023). A systematic review and meta-analysis of the omega-3 fatty acids effects on brain-derived neurotrophic factor (BDNF). Nutritional Neuroscience, 27(7), 715–725. https://doi.org/10.1080/1028415x.2023.2245996
-
- Hoffer, M. E., Balaban, C., Slade, M. D., Tsao, J. W., & Hoffer, B. (2013). Amelioration of acute sequelae of blast induced mild traumatic brain injury by N-acetyl cysteine: A double-blind, placebo controlled study. PLoS ONE, 8(1). https://doi.org/10.1371/journal.pone.0054163
-
- Aoyama, K., & Nakaki, T. (2013). Impaired glutathione synthesis in neurodegeneration. International Journal of Molecular Sciences, 14(10), 21021–21044. https://doi.org/10.3390/ijms141021021
-
- Roth, T. L., Nayak, D., Atanasijevic, T., Koretsky, A. P., Latour, L. L., & McGavern, D. B. (2014). Transcranial amelioration of inflammation and cell death after brain injury. Nature, 505(7482), 223–228. https://doi.org/10.1038/nature12808
-
- Laird, M. D., Sukumari‐Ramesh, S., Swift, A. E., Meiler, S. E., Vender, J. R., & Dhandapani, K. M. (2010). Curcumin attenuates cerebral edema following traumatic brain injury in mice: A possible role for aquaporin‐4? Journal of Neurochemistry, 113(3), 637–648. https://doi.org/10.1111/j.1471-4159.2010.06630.x
-
- Khayatan, D., Razavi, S. M., Arab, Z. N., Niknejad, A. H., Nouri, K., Momtaz, S., Gumpricht, E., Jamialahmadi, T., Abdolghaffari, A. H., Barreto, G. E., & Sahebkar, A. (2022). Protective effects of curcumin against traumatic brain injury. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 154, 113621. https://doi.org/10.1016/j.biopha.2022.113621
-
- Garodia, P., Hegde, M., Kunnumakkara, A. B., & Aggarwal, B. B. (2023). Curcumin, inflammation, and neurological disorders: How are they linked?. Integrative medicine research, 12(3), 100968. https://doi.org/10.1016/j.imr.2023.100968
-
- Zhang, M., Hao, Z., Wu, J., Teng, Z., Qiu, W., & Cheng, J. (2023). Curcumin ameliorates traumatic brain injury via C1ql3-mediated microglia M2 polarization. Tissue & cell, 84, 102164. https://doi.org/10.1016/j.tice.2023.102164
-
- Razmkon, A., Sadidi, A., Sherafat-Kazemzadeh, E., Mehrafshan, A., Jamali, M., Malekpour, B., & Saghafinia, M. (2011). Administration of Vitamin C and vitamin E in severe head injury: A randomized double-blind controlled trial. Neurosurgery, 58, 133–137. https://doi.org/10.1227/neu.0b013e3182279a8f
-
- Marx, C. E., Naylor, J. C., Kilts, J. D., Dunn, C. E., Tupler, L. A., Szabo, S. T., Capehart, B. P., Morey, R. A., Shampine, L. J., & Acheson, S. K. (2016). Neurosteroids and Traumatic Brain Injury: Translating Biomarkers to Therapeutics; Overview and Pilot Investigations in Iraq and Afghanistan Era Veterans. In D. Laskowitz (Eds.) et. al., Translational Research in Traumatic Brain Injury. CRC Press/Taylor and Francis Group.
-
- Arora, N., Shastri, D. H., Patel, U. P., & Bhatia, K. (2024). Modulation of beta-hydroxybutyrate in traumatic brain injury. Current opinion in clinical nutrition and metabolic care, 27(2), 168–177. https://doi.org/10.1097/MCO.0000000000001008
-
- Davis, L. M., Pauly, J. R., Readnower, R. D., Rho, J. M., & Sullivan, P. G. (2008). Fasting is neuroprotective following traumatic brain injury. Journal of Neuroscience Research, 86(8), 1812–1822. https://doi.org/10.1002/jnr.21628
-
- Daines, S. A. (2021). The therapeutic potential and limitations of ketones in traumatic brain injury. Frontiers in Neurology, 12. https://doi.org/10.3389/fneur.2021.723148
-
- Llorente-Folch, I., Düssmann, H., Watters, O., Connolly, N. M., & Prehn, J. H. (2023). Ketone body β-hydroxybutyrate (BHB) preserves mitochondrial bioenergetics. Scientific Reports, 13(1). https://doi.org/10.1038/s41598-023-46776-8
-
- Jensen, N. J., Wodschow, H. Z., Nilsson, M., & Rungby, J. (2020). Effects of ketone bodies on brain metabolism and function in neurodegenerative diseases. International Journal of Molecular Sciences, 21(22), 8767. https://doi.org/10.3390/ijms21228767
-
- Hiskens, M. I., Li, K. M., Schneiders, A. G., & Fenning, A. S. (2023). Repetitive mild traumatic brain injury-induced neurodegeneration and inflammation is attenuated by acetyl-l-carnitine in a preclinical model. Frontiers in Pharmacology, 14. https://doi.org/10.3389/fphar.2023.1254382
-
- Tashakori-Miyanroudi, M., Ramazi, S., Hashemi, P., Nazari-Serenjeh, M., Baluchnejadmojarad, T., & Roghani, M. (2022). Acetyl-L-Carnitine Exerts Neuroprotective and Anticonvulsant Effect in Kainate Murine Model of Temporal Lobe Epilepsy. Journal of molecular neuroscience : MN, 72(6), 1224–1233. https://doi.org/10.1007/s12031-022-01999-8
-
- Meshkini, A., Meshkini , M., & Sadeghi-Bazargani, H. (2016). Citicoline for Traumatic Brain Injury: A systematic review & meta-analysis. Journal of Injury and Violence Research, 9(1). https://doi.org/10.5249/jivr.v9i1.843
-
- Secades, J. J. (2021). Role of citicoline in the management of Traumatic Brain Injury. Pharmaceuticals, 14(5), 410. https://doi.org/10.3390/ph14050410
-
- Watanabe, S., Kono, S., Nakashima, Y., Mitsunobu, K., & Otsuki, S. (1975). Effects of various cerebral metabolic activators on glucose metabolism of brain. Psychiatry and Clinical Neurosciences, 29(1), 67–76. https://doi.org/10.1111/j.1440-1819.1975.tb02324.x
-
- Krishna, D., Chaurasia, I.D., & Jethwani, U. (2012). ROLE OF CITICOLINE IN TRAUMATIC BRAIN INJURY: A RANDOMIZED CONTROLLED STUDY. Journal of Medicine.
-
- Trimmel, H., Majdan, M., Wodak, A., Herzer, G., Csomor, D., & Brazinova, A. (2018). Citicoline in severe traumatic brain injury: indications for improved outcome : A retrospective matched pair analysis from 14 Austrian trauma centers. Wiener klinische Wochenschrift, 130(1-2), 37–44. https://doi.org/10.1007/s00508-017-1240-9
-
- Zafonte, R., Friedewald, W. T., Lee, S. M., Levin, B., Diaz-Arrastia, R., Ansel, B., Eisenberg, H., Timmons, S. D., Temkin, N., Novack, T., Ricker, J., Merchant, R., & Jallo, J. (2009). The citicoline brain injury treatment (COBRIT) trial: design and methods. Journal of neurotrauma, 26(12), 2207–2216. https://doi.org/10.1089/neu.2009.1015
-
- Vink R. (2016). Magnesium in the CNS: recent advances and developments. Magnesium research, 29(3), 95–101. https://doi.org/10.1684/mrh.2016.0408
-
- Busingye, D. S., Turner, R. J., & Vink, R. (2016). Combined magnesium/polyethylene glycol facilitates the neuroprotective effects of magnesium in traumatic brain injury at a reduced magnesium dose. CNS Neuroscience & Therapeutics, 22(10), 854–859. https://doi.org/10.1111/cns.12591
-
- Kim, Y.-S., Won, Y. J., Lim, B. G., Min, T. J., Kim, Y.-H., & Lee, I. O. (2020). Neuroprotective effects of magnesium L-threonate in a hypoxic zebrafish model. BMC Neuroscience, 21(1). https://doi.org/10.1186/s12868-020-00580-6
-
- Hoane M. R. (2004). Magnesium therapy and recovery of function in experimental models of brain injury and neurodegenerative disease. Clinical calcium, 14(8), 65–70.
-
- Enomoto, T., Noda, Y., & Nabeshima, T. (2004). Neuroprotective effects of magnesium on cerebral ischemia and cerebral contusion. Clinical calcium, 14(8), 60–64.
-
- Hoane M. R. (2007). Assessment of cognitive function following magnesium therapy in the traumatically injured brain. Magnesium research, 20(4), 229–236.
-
- Young, J.M., & Hoane, M.R. (2018). administration after experimental traumatic brain injury improves decision-making skills. Brain Research Bulletin, 139, 182-189.
-
- Pérez-Sala, D., & Pajares, M. A. (2023). Appraising the Role of Astrocytes as Suppliers of Neuronal Glutathione Precursors. International journal of molecular sciences, 24(9), 8059. https://doi.org/10.3390/ijms24098059
-
- Wojcik, M., Burzynska-Pedziwiatr, I., & Wozniak, L. (2010). A review of natural and synthetic antioxidants important for health and longevity. Current Medicinal Chemistry, 17(28), 3262–3288. https://doi.org/10.2174/092986710792231950
-
- Forbes, S. C., Cordingley, D. M., Cornish, S. M., Gualano, B., Roschel, H., Ostojic, S. M., Rawson, E. S., Roy, B. D., Prokopidis, K., Giannos, P., & Candow, D. G. (2022). Effects of creatine supplementation on Brain Function and Health. Nutrients, 14(5), 921. https://doi.org/10.3390/nu14050921
-
- Roschel, H., Gualano, B., Ostojic, S. M., & Rawson, E. S. (2021). Creatine supplementation and Brain Health. Nutrients, 13(2), 586. https://doi.org/10.3390/nu13020586
-
- Newman, J. M., Pekari, T. B., & Van Wyck, D. W. (2023). Neuroprotection and Therapeutic Implications of Creatine Supplementation for Brain Injury Complications. Medical journal (Fort Sam Houston, Tex.), (Per 23-4/5/6), 31–38.
-
- Amlerova, Z., Chmelova, M., Anderova, M., & Vargova, L. (2024). Reactive gliosis in traumatic brain injury: a comprehensive review. Frontiers in cellular neuroscience, 18, 1335849. https://doi.org/10.3389/fncel.2024.1335849
-
- Bailes, J. E., Dashnaw, M. L., Petraglia, A. L., & Turner, R. C. (2014). Cumulative effects of repetitive mild traumatic brain injury. Progress in neurological surgery, 28, 50–62. https://doi.org/10.1159/000358765
-
- Cherian, K. N., Keynan, J. N., Anker, L., Faerman, A., Brown, R. E., Shamma, A., Keynan, O., Coetzee, J. P., Batail, J.-M., Phillips, A., Bassano, N. J., Sahlem, G. L., Inzunza, J., Millar, T., Dickinson, J., Rolle, C. E., Keller, J., Adamson, M., Kratter, I. H., & Williams, N. R. (2024). Magnesium–ibogaine therapy in veterans with traumatic brain injuries. Nature Medicine, 30(2), 373–381. https://doi.org/10.1038/s41591-023-02705-w
-
- Cieri MB, Ramos AJ. (2025). Astrocytes, reactive astrogliosis, and glial scar formation in traumatic brain injury. Neural Regen Res, 20(4):973-989. DOI: 10.4103/NRR.NRR-D-23-02091.
-
- Jamjoom, A. A. B., Rhodes, J., Andrews, P. J. D., & Grant, S. G. N. (2021). The synapse in traumatic brain injury. Brain, 144(18–31). doi:10.1093/brain/awaa321
